
Garden and Plate
The Molecular Biology of Nutrition










|
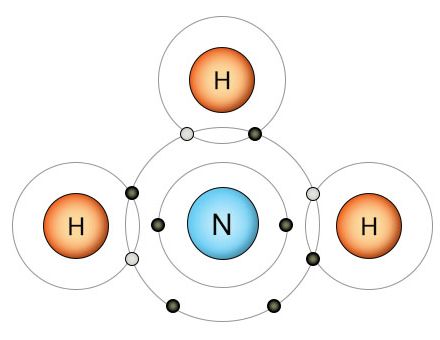
Nitrogenase breaks the triple bonds of the N2 molecule and replaces each bond with a hydrogen atom. This results in the ammonia molecule above. A proton (H+) takes one of ammonia's two free electrons to become a hydrogen atom, then forms a fourth covalent bond with the last free electron. This creates the ammonium ion we see below. 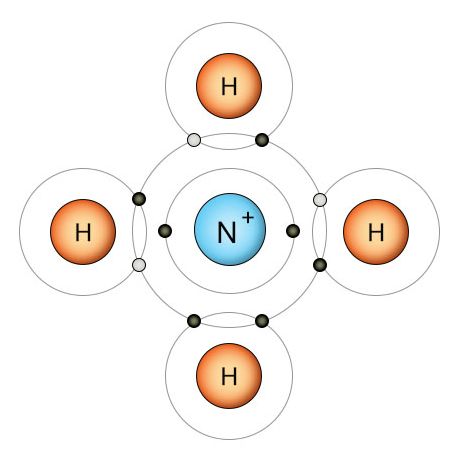
|
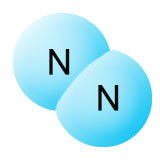
The breaking apart of paired up nitrogen (N2) molecules is referred to as nitrogen fixation. Only a few kinds of bacteria (the diazotrophs) produce the nitrogenase enzymes that enable them to do this. Plants and animals don't produce these enzymes. There's more than one type of diazotroph, but the most common variety in your garden are the rhizobia. They exist in a symbiotic relationship with the roots of legumes, providing them with fixed nitrogen in return for carbohydrates. The legume family includes plants like peas, beans, clover, soy, and alfalfa.
Once the nitrogenase enzyme breaks the three covalent bonds that hold the N2 molecule together, it fills the three newly available empty slots with hydrogen atoms. If this doesn't take place, the nitrogen atoms will recombine into N2. Once the hydrogen is in place, we have an ammonia (NH3) molecule (shown on the upper right). The nitrogen atom now has three of it's outer electrons bound up in covalent bonds with hydrogen atoms, and two additional unbound electrons. It has a full outer shell, so it doesn't need any more electrons. The watery soup it's floating in, however, is full of easily accessible protons. A proton (H+) is a hydrogen atom that's missing it's electron. Protons (hydrogen ions) separate from water molecules easily due to oxygen's strong pull on hydrogen's electron. When the proton finds the ammonia molecule, it attaches to one of nitrogen's two unbound electrons and uses it to form a covalent bond with nitrogen's remaining unbound electron. This creates the ammonium ion (NH4+) shown on the lower right.

Peas are a part of the legume family. They form a symbiotic relationship with rhizobia bacteria that live on their roots. The bacteria fix nitrogen for the pea plant, and the pea plant gives them carbohydrates in return. |
The ammonium ion is biologically useful, because the relatively weak nitrogen-hydrogen covalent bonds serve as placeholders. They can be easily broken when the nitrogen is needed for other purposes. Once the diazotrophs have converted the N2 molecules into ammonium ions, they become fair game for other organisms. Some soil bacteria replace the hydrogen atoms with oxygen atoms, creating nitrates (NO3-). The nitrates and ammonium ions can both be absorbed by plant roots. The plant then converts the nitrates back into ammonium ions, and uses the ammonium ions to create amino and nucleic acids.
While most naturally produced free nitrogen originally comes from the activities of rhizobia bacteria, most plants don't have a symbiotic relationship with these organisms. Outside of the legumes, most plants get all of their fixed nitrogen secondhand. The bacteria fix the nitrogen just once, then it's recycled over and over again, moving from plant to animal and back again in a process called the nitrogen cycle. Animals eat the plants, then return the nitrogen to the soil as manure. When plants decompose back down into compost or manure, the nitrogen is made available to subsequent plants. In this way, the nitrogen initially created by the rhizobia bacteria eventually builds up in the soil, even though it's being taken up by the plants and animals.
The exception to this is when plants are removed from the environment. When we farm or garden, plants take up nutrients from the soil. We then remove the plants for food and use the nutrients for ourselves. If we don't put the nutrients back into the soil, they get depleted faster than nature can replenish them. That's why we have to keep adding compost, manure, or chemical fertilizers to our farms and gardens. Chlorophyll molecules, which play a central role in photosynthesis, use lots of nitrogen. That's why adding nitrogen to the garden increases plant growth, especially in the green, chlorophyll packed leaves.